Research Direction Introduction
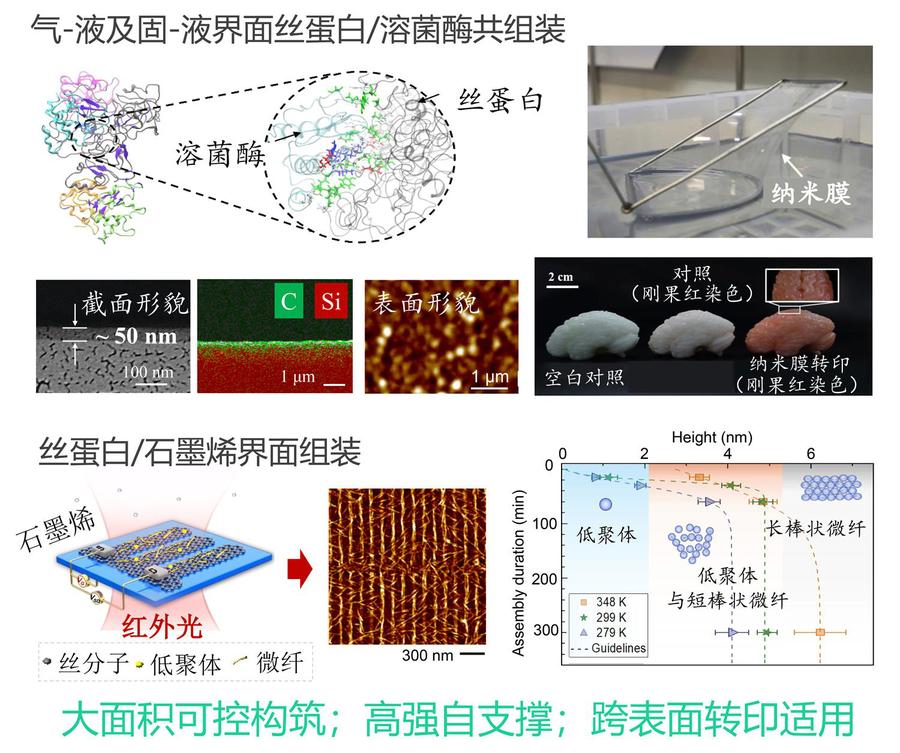
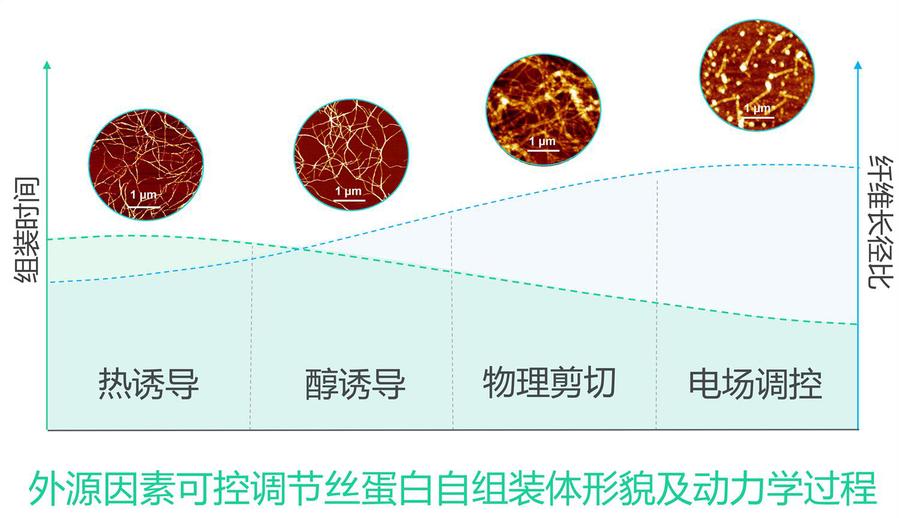
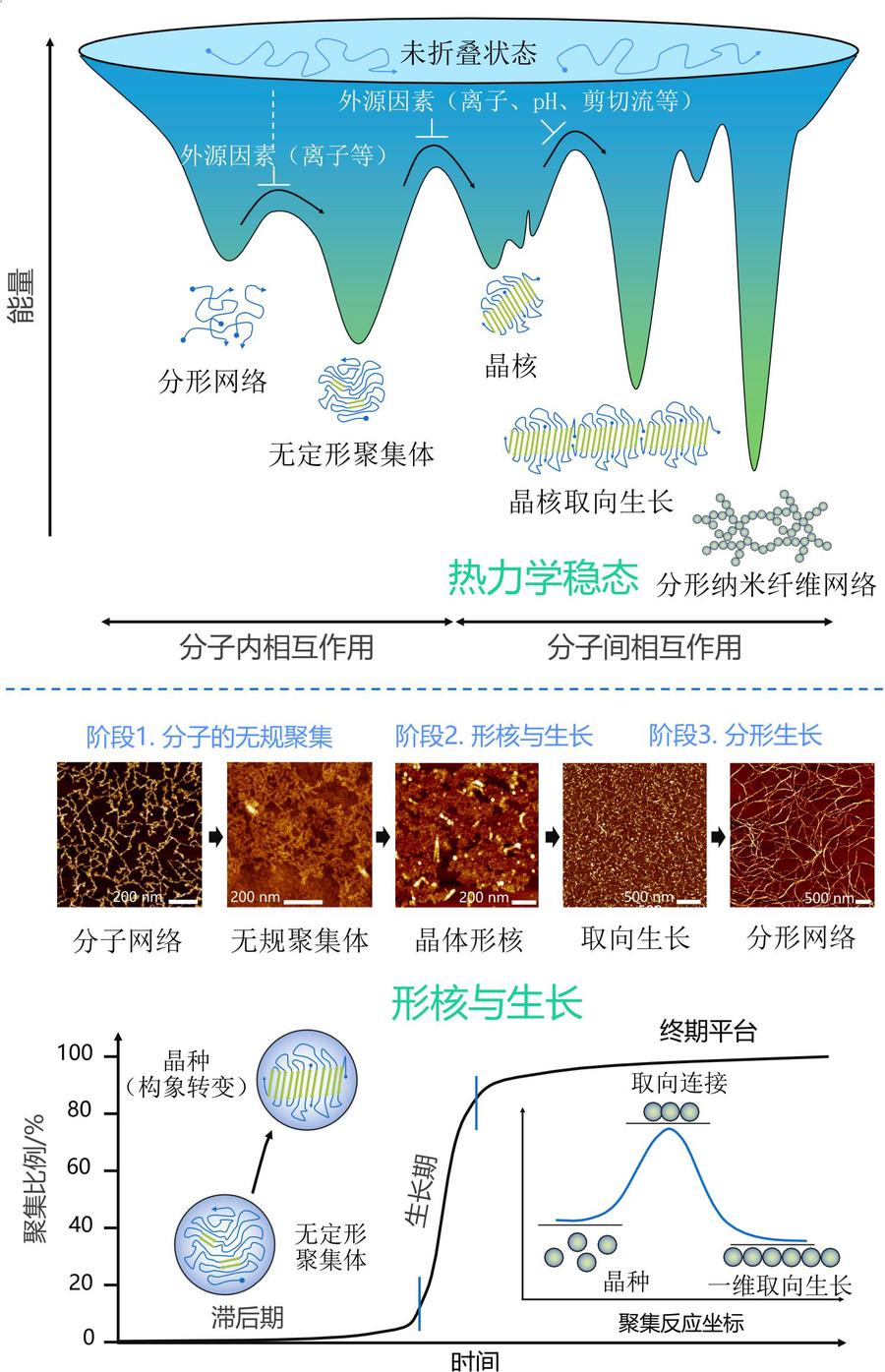
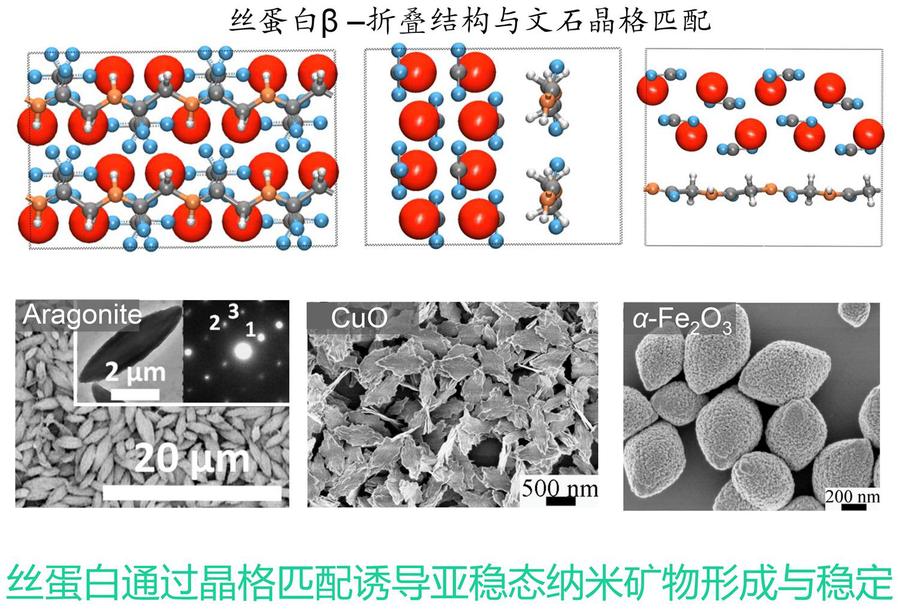
The alternating arrangement of hydrophilic-hydrophobic domains in silk fibroin endows it with an inherent self-assembly tendency, with the assembly process regulated by nucleation-growth kinetics. Various exogenous factors can precisely control the morphology of assemblies (such as microfibrils and microspheres) and kinetic pathways. Its β-sheet structure provides biomimetic mineralization templates for inorganic minerals, inducing the formation of metastable crystal phases, thereby expanding preparation strategies for biomedical materials (such as bone repair) and new energy materials (such as electrodes). Furthermore, silk fibroin with lysozyme and other proteins can form dense, self-supporting ultrathin films at gas-liquid/solid-liquid interfaces with transferable properties, providing novel structural building blocks for the development of micro-nano functional devices.
Silk protein self-assembly
Silk protein assembly thermal/kinetic mechanism
Biomineralization
interface assembly