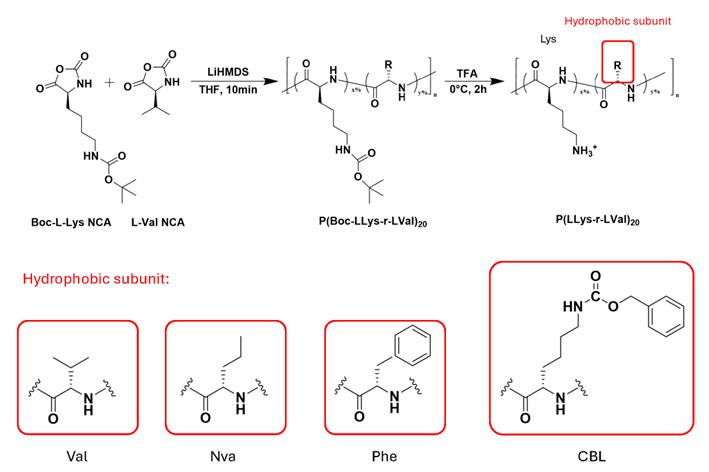
Bacterial biofilms shield embedded cells behind extracellular polymeric substance (EPS) barriers, frustrate antibiotic penetration, and protect persisters, demanding materials that couple broad antimicrobial activity with efficient biofilm clearance. We establish a design framework for host defense peptide (HDP) mimics using poly(α-amino acid) copolymers with a cationic lysine backbone (degree of polymerization ≈ 20) and tunable hydrophobic modules—valine (Val), norvaline (Nva), phenylalanine (Phe), and N-ε-carbobenzoxy-lysine (CBL). Random ring-opening copolymerization of α-amino acid N-carboxyanhydride (NCA) (0-60% hydrophobic feed) yields amphiphilic libraries whose composition programs secondary structure: Val biases β-sheet formation, high Nva favors α-helices, Phe promotes ordered states via π-π stacking, and CBL side-chain amides build stabilizing hydrogen-bond networks. Across MIC/MBC testing, S. aureus and P. aeruginosa exhibit distinct requirements: cationic charge density is the primary determinant for penetrating EPS and clearing mature Gram-positive biofilms, whereas moderate hydrophobicity—notably Nva and CBL—optimizes activity against Gram-negative pathogens by balancing solubility, membrane affinity, and EPS interactions. Self-assembly refines performance further; nanoscale aggregates with elevated surface potential (e.g., Lys60CBL40) deepen penetration and accelerate persister eradication. Critically, antibacterial potency against planktonic cells does not predict antibiofilm efficacy, revealing mechanistic decoupling that necessitates independent tuning. These results deliver a quantitative structure-function map and actionable rules for engineering amphiphilic polymers that recapitulate the functional hallmarks of HDPs, enabling simultaneous planktonic killing and robust clearance of mature biofilms, and offering promising candidates for multidrug-resistant, biofilm-associated infections.